What a difference one halogen atom can make! Fentanyl is a very powerful opioid painkiller usually reserved for cancer patients and others with acute and intractable pain. Recently, fentanyl has been found in samples of heroin sold on the street. Last year a man overdosed in Randolph after injecting what he thought was heroin. It was largely fentanyl. Fentanyl is so powerful, and so dangerous, because it is 50 - 100 times more powerful than morphine. Fentanyl affects all tissues in the body and overdoses cause depression of the respiratory system, and cause breathing to stop. In addition, fentanyl, like other opioids, is highly addictive.
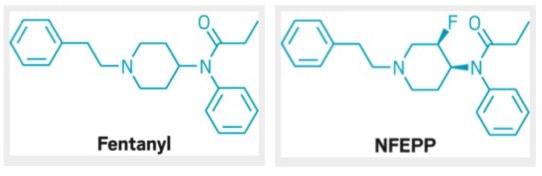
Last week researchers from Berlin published a potential solution in Science magazine. The work is summarized in this excellent article in Ars Technica. The researchers added a fluorine atom to a ring proximal to the tertiary amine group of fentanyl, creating a molecule called N-(3-fluoro-1-phenethylpiperidin-4-yl)-N-phenyl proionamide (NFEPP). The fluorine pulled electron density away from the neighboring nitrogen decreasing its ability to form a dative bond with hydrogens from its environment. This caused the pka of the NFEPP derivative to be lower (6.8) than that of fentanyl (8.4). So, NFEPP becomes protonated only in regions of the body that are quite acidic. Why is that important? It turns out that areas of inflammation are more acidic (pH 5 - 7) than surrounding tissue (pH 7.4). Fentanyl and NFEPP are only effective when their tertiary amine groups are protonated. So, unlike fentanyl, NFEPP is only active in areas of the body that are inflamed; it is essentially a targeted painkiller! When tested in mice, NFEPP doesn't cause respiratory distress, overdose and doesn't appear to cause the 'reward seeking behavior' associated with addiction. Go fluorine!
 RSS Feed
RSS Feed