|
Cattle and other ruminants create methane as they digest their food. And when the belch and fart they add pretty amazing amounts of this greenhouse gas that is 27-times as 'warming' as carbon dioxide. Cows are responsible for 14.5% of all greenhouse gases. Now scientists at the University of Guelph in Canada have discovered that cow genetics help determine how much methane cows produce. Follow-up studies will try to breed 'low-methane' cows. For more on this study, connect with the NPR report or go right to the paper in PLOS Genetics.
0 Comments
A new study from MIT's Sloan School of Management and the University of Pittsburgh finds that scientists around the world are relying on Wikipedia to access scientific information and research. Wikipedia is the 5th most used site on the web! A report about the study shows that information from Wikipedia, and scientists' growing reliance on this information, is shaping scientific research, influencing one word out of every three hundred words published.
I feel vindicated as I encourage my students to use Wikipedia as a resource for studying science. Of course, I'm a strong believer in using the site as a route to primary sources. Recently, innovative catalysts have allowed scientists to convert carbon dioxide into single carbon fuels like methanol or formate, but creating multi-carbon fuels has been tougher. Using catalysts to build two- or three-carbon fuels has required high voltage electricity and hasn't been energy efficient. But now, chemists at UC Berkeley have created a catalyst by depositing copper nanoparticles onto carbon paper, and this catalyst works with electricity that can be produced by solar panels. This new catalyst can produce ethylene, ethanol and propanol when supplied with carbon dioxide and 600 millivolts of electricity.
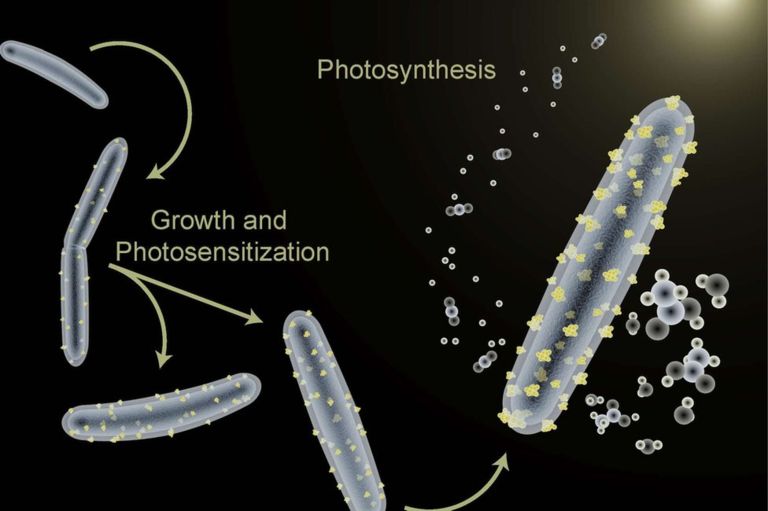
This type of technology could allow us to recycle the carbon dioxide released by combustion of traditional fuels into 'new' fuel via a primitive form of artificial photosynthesis. And recycling carbon dioxide should help us begin to control global greenhouse gas concentrations and global climate change! Read more here. Scientists at UC Berkeley have exploited the ability of some strains of bacteria to defend themselves against harmful heavy metals to create bacteria that use sunlight to make acetic acid.
Get more information via this link to the BBC's science page. At the beginning of each semester I do a chemical safety training for my chemistry students. I always mention the danger of peroxides, particularly when the mix with organics. Well, when hurricane Harvey hit Crosby, Texas, six feet of flood water disabled the safe, refrigerated storage of organic peroxides at the Arkema chemicals plant. Plant chemists and administrators warned that the chemicals would become unstable as they warmed and that explosion would occur. And this morning it did. More explosions are coming.
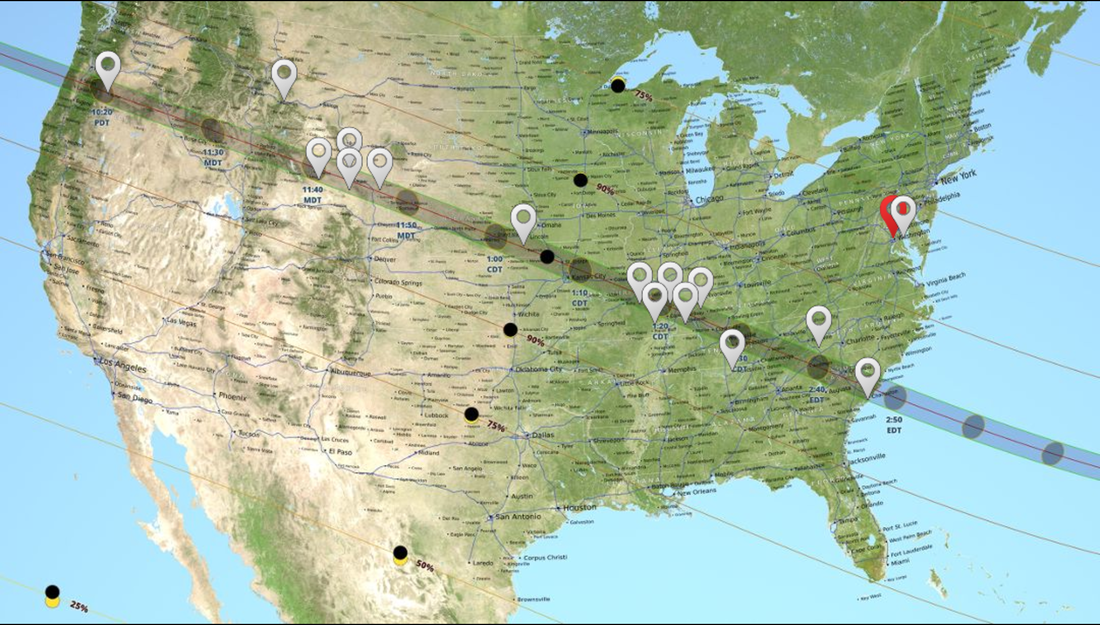
The Washington Post reported the story today. Is there a hidden meaning to all this? Well possibly, but that's far above my pay grade! In any case, a total eclipse is a rare and splendid thing that we are privileged to see. In Vermont we won't see a total eclipse, but what appears to be a partial eclipse as we aren't directly below the sun's line of travel over the earth. To learn more about the eclipse check out this animation of the eclipse in Vermont. Or have a look at my brother's great presentation on eclipses. My brother, Michael Richmond, is an astrophysicist who teaches at Rochester Institute of Technology where he is also the director of the observatory. He's watching the eclipse in Nashville, TN where he's serving as the eclipse expert at a Kimpton Hotel. Good deal for everyone! Path of today's eclipse, courtesy of NASA.
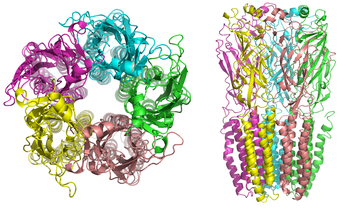
Caribbean cone snails (Conus regius) kill prey, including small fish, by harpooning them with hollow barb that pumps a complex mixture of at least ten neurotoxins into their prey. Researchers at the University of Utah have just published in PNAS, and their work is summarized by phys.org. A small protein, Rg1A, binds to the nicotinic acetylcholine receptor and blocks chronic pain for 72 hours after it is injected into rodents. This is a non-opioid pathway, suggesting that there may be new, and less dangerous ways to treat pain. Researchers then did something incredibly clever. The synthesized 20 variants of the Rg1A peptide and found one, Rg1A4, that binds to the human nicotinic acetylcholine receptor. They went back to the rodent model to confirm that Rg1A4 prevented pain and did so by blocking the receptor, preventing it from sending pain signals. This new pain-blocking pathway will be the focus of much additional research.
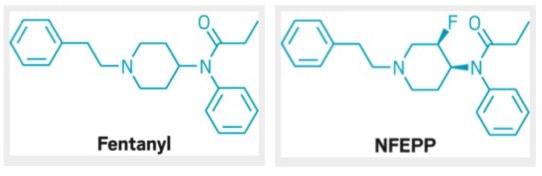
(1) By Ataly (Own work) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0) or GFDL (http://www.gnu.org/copyleft/fdl.html)], via Wikimedia Commons (2) https://nihrecord.nih.gov/newsletters/2005/03_01_2005/story03.htm What a difference one halogen atom can make! Fentanyl is a very powerful opioid painkiller usually reserved for cancer patients and others with acute and intractable pain. Recently, fentanyl has been found in samples of heroin sold on the street. Last year a man overdosed in Randolph after injecting what he thought was heroin. It was largely fentanyl. Fentanyl is so powerful, and so dangerous, because it is 50 - 100 times more powerful than morphine. Fentanyl affects all tissues in the body and overdoses cause depression of the respiratory system, and cause breathing to stop. In addition, fentanyl, like other opioids, is highly addictive. Last week researchers from Berlin published a potential solution in Science magazine. The work is summarized in this excellent article in Ars Technica. The researchers added a fluorine atom to a ring proximal to the tertiary amine group of fentanyl, creating a molecule called N-(3-fluoro-1-phenethylpiperidin-4-yl)-N-phenyl proionamide (NFEPP). The fluorine pulled electron density away from the neighboring nitrogen decreasing its ability to form a dative bond with hydrogens from its environment. This caused the pka of the NFEPP derivative to be lower (6.8) than that of fentanyl (8.4). So, NFEPP becomes protonated only in regions of the body that are quite acidic. Why is that important? It turns out that areas of inflammation are more acidic (pH 5 - 7) than surrounding tissue (pH 7.4). Fentanyl and NFEPP are only effective when their tertiary amine groups are protonated. So, unlike fentanyl, NFEPP is only active in areas of the body that are inflamed; it is essentially a targeted painkiller! When tested in mice, NFEPP doesn't cause respiratory distress, overdose and doesn't appear to cause the 'reward seeking behavior' associated with addiction. Go fluorine!
You've probably never heard of Hans Rosling, but he was the rock star of statistics presentation. Sound oxymoronic? When I stumbled across his work a few years ago I was reminded that presentation is nine-tenths of the law. How you tell a story is often just as important as the story itself, and Hans Rosling pulled the human stories out of piles of data and made them sing with his Gapmider software. The NPR story links to videos of some of his best presentations. Oh, and he was also an amateur sword swallower (see end of this video).
Peace Hans. As a former vegetarian I love this idea! A professor at Stanford University realized that raising meat is costly to the environment and may not be able to feed our rapidly growing human population. At the same time, he realized that humans have been eating meet for thousands of years and love its taste, and that most 'veggie burgers' just don't deliver on taste. So, he found that much of the taste of a burger comes from the hemoglobin, or blood protein, in the meet. Turns out that plants make small amounts of hemoglobin too. Brown's lab engineered the gene for vegetable hemoglobin into yeast to produce large amounts of it. The resulting burger looks and tastes pretty authentic:
"To charge a premium, Duff says, the makers will have to work hard to tell the story that this burger is better for the Earth, because he doesn't think the taste alone will stand out for people. If you never told people what's in the burger, Duff says, "they quite literally would not know." |
About meI'm a professor of science working in the beautiful heart of rural Vermont. Archives
December 2017
Categories |






 RSS Feed
RSS Feed